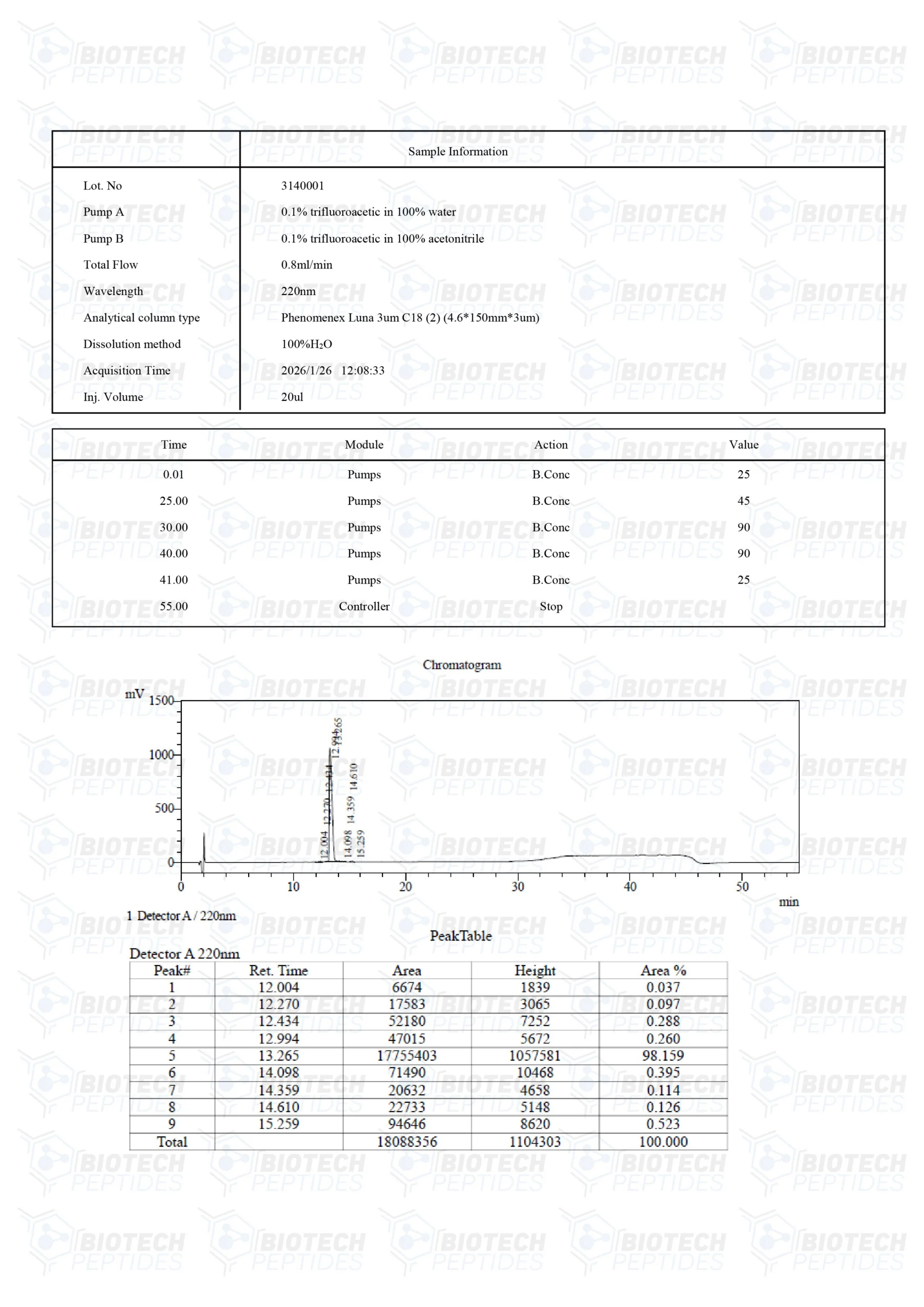
ACE-031 (1mg)
$161.00
ACE-031 peptides are Synthesized and Lyophilized in the USA.
Discount per Quantity
| Quantity | 5 - 9 | 10 + |
|---|---|---|
| Discount | 5% | 10% |
| Price | $152.95 | $144.90 |
FREE - USPS priority shipping
ACE-031 Peptide
ACE-031 is a soluble protein comprising the type IIB activin receptor (ACVR2B) and the immunoglobulin G1-Fc (IgG1-Fc), and has been studied for its potential in binding and inactivating myostatin, a protein that hampers muscle cell growth. It has been examined during the course of various research studies involved in muscle-wasting disorders and neuromuscular conditions such as Duchenne Muscular Dystrophy (DMD), as well as certain cancers such as those of the prostate or colorectal regions and lipid storage and bone metabolism.
Specifications
Other Known Titles: Ramatercept, ActRIIB-IgG1, ACVR2B/Fc
PubChem: CID 16130571
ACE-031 Research
ACE-031 and Muscle Cells
ACE-031 appears to enhance muscle growth and bone metabolism; it also may limit lipid accumulation. A notable improvement in lean body mass and thigh muscle volume was observed in experimental studies within a month of ACE-031 exposure.[1] The outcome has the potential added benefits of improved bone and fat metabolic profile. Research is ongoing. ACE-031 introduction also appears to reduce muscle wasting through its potential to bind myostatin in muscle cells.[2] ACE-031, while not a commonly researched peptide compound, has been posited by researchers to elicit synergistic impact alongside certain growth hormone analogs, like IGF-1.
ACE-031 and Energy Metabolism
Research has suggested that inhibiting the natural ACE-031 proteins may fail to reduce serum lactate levels, preventing ultimate metabolic damage to muscles, and vascularization of the muscle tissue. These impacts may potentially be mitigated through ACE-031 supplementation. It has been speculated to promote muscle cell growth by blocking myostatin-mediated wasting and may delay the onset of fatigue and oxidative damage by enhancing the oxygenation of the muscle tissues.
ACE-031 and Muscle Force
ACE-031 has been studied for its potential influence in muscle function. Other than blocking the activity of myostatin, ACE-031 has been researched for its possible ability to improve muscle thermodynamics by promoting oxidative respiration, thereby improving the force-generating capacity of muscles, specifically the maximum force by 50% and total contractile force by 25%.[3] Experimental studies have suggested that ACE-031 may improve muscle strength potentially without affecting energy dynamics, or changes in ATP levels or contractile efficiency within the muscle tissues.
ACE-031 and Muscle Repair
Muscle wasting disorders such as Duchenne Muscular Dystrophy (DMD) often result in physical difficulty. Research models of the disorder display severe muscle loss due to low protein levels despite the extremely high-fat reserves—the primary reason being that the dystrophin (a group of proteins) in these models appears to be non-functional. Moreover, the release of myostatin from damaged muscle cells may affect surrounding cells causing their growth to slow. However, research has suggested the potential of ACE-031 to reduce the rate of muscle damage by addressing the impacts of myostatin release.[4] The peptide appears to preserve muscle function, increase lean body mass, improve bone mineral density, and reduce fat reserves. Research is ongoing.
ACE-031 and Bone Density
Researchers observed that ACE-031 appeared to improve total body weight, muscle mass, and bone mineral density when introduced to an experimental research model once a week for seven consecutive weeks.[5] A reduction in the population of osteoclasts appeared to be responsible for the improved mineral content in bones, which also apparently improved the biomechanics, stiffness, and maximum force that the bones could tolerate. Research has suggested the potential of ACE-031 to increase bone mass by around 30%, positing its impact in controlling the progression of osteoporosis. In addition to its possible myostatin inhibiting characteristics, studies have suggested the ability of ACE-031 to increase bone density by almost 132% in the femur (the thigh bone, which is often damaged with age) and 27% in vertebrae.
ACE-031 and Cancer, Muscle Loss
Molecular cascades resulting in muscle loss due to either death or necrosis are commonly seen in research associated with cancer cells. The primary reason is the metabolic stress on muscles due to changes in aerobic respiration status. In addition, there is an increase in the free radical population within cells that indirectly causes muscle damage. Activating the ERK1/2 pathway by introducing ACE-031 appears to avert muscle fiber atrophy due to apoptosis. In addition, the efficiency of energy consumption and the mitochondrial metabolism was also observed to improve in research studies. Moreover, the concentration of free radicals also appeared to reduce. Furthermore, myostatin is produced in certain cancers, which may lead to muscle wasting and loss. In addition, these transformed cells are often associated with inactivated activin receptors, loss of mitochondria, and hence ATP levels. Research has suggested a reversal of these actions with ACE-031 exposure. Some additional potential impacts associated with myostatin inhibition include improved insulin sensitivity, reduced fat storage, reduced inflammation, and better bone metabolism and strength.
Disclaimer: The products mentioned are not intended for human or animal consumption. Research chemicals are intended solely for laboratory experimentation and/or in-vitro testing. Bodily introduction of any sort is strictly prohibited by law. All purchases are limited to licensed researchers and/or qualified professionals. All information shared in this article is for educational purposes only.
References
- Attie, K. M., Borgstein, N. G., Yang, Y., Condon, C. H., Wilson, D. M., Pearsall, A. E., Kumar, R., Willins, D. A., Seehra, J. S., & Sherman, M. L. (2013). A single ascending-dose study of muscle regulator ACE-031 in healthy volunteers. Muscle & nerve, 47(3), 416–423. https://doi.org/10.1002/mus.23539
- Cadena, S. M., Tomkinson, K. N., Monnell, T. E., Spaits, M. S., Kumar, R., Underwood, K. W., Pearsall, R. S., & Lachey, J. L. (2010). Administration of a soluble activin type IIB receptor promotes skeletal muscle growth independent of fiber type. Journal of applied physiology (Bethesda, Md. : 1985), 109(3), 635–642. https://doi.org/10.1152/japplphysiol.00866.2009
- Muramatsu, H., Kuramochi, T., Katada, H., Ueyama, A., Ruike, Y., Ohmine, K., Shida-Kawazoe, M., Miyano-Nishizawa, R., Shimizu, Y., Okuda, M., Hori, Y., Hayashi, M., Haraya, K., Ban, N., Nonaka, T., Honda, M., Kitamura, H., Hattori, K., Kitazawa, T., Igawa, T., … Nezu, J. (2021). Novel myostatin-specific antibody enhances muscle strength in muscle disease models. Scientific reports, 11(1), 2160. https://doi.org/10.1038/s41598-021-81669-8
- Campbell, C., McMillan, H. J., Mah, J. K., Tarnopolsky, M., Selby, K., McClure, T., Wilson, D. M., Sherman, M. L., Escolar, D., & Attie, K. M. (2017). Myostatin inhibitor ACE-031 treatment of ambulatory boys with Duchenne muscular dystrophy: Results of a randomized, placebo-controlled clinical trial. Muscle & nerve, 55(4), 458–464. https://doi.org/10.1002/mus.25268
- Maïmoun, L., Mariano-Goulart, D., Huguet, H., Renard, E., Lefebvre, P., Picot, M. C., Dupuy, A. M., Cristol, J. P., Courtet, P., Boudousq, V., Avignon, A., Guillaume, S., & Sultan, A. (2022). In patients with anorexia nervosa, myokine levels are altered but are not associated with bone mineral density loss and bone turnover alteration. Endocrine connections, 11(5), e210488. https://doi.org/10.1530/EC-21-0488